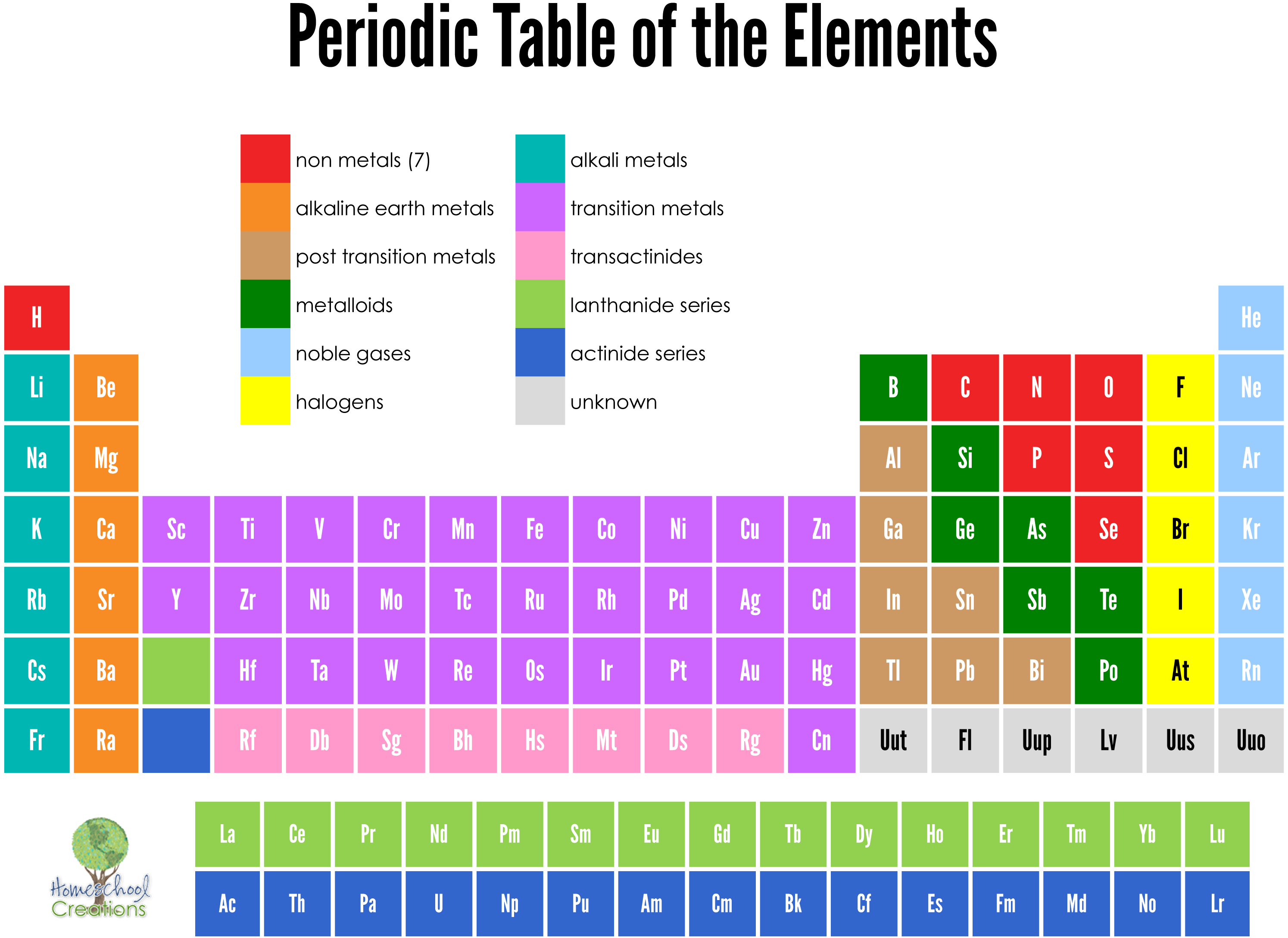
The periodic table can be a little tricky to read, but the more you learn, the better you will be able to do in the sciences. A metal has a solid or malleable substance while a non-metal is a gas at room temperature. In the middle of the table are the Rare Earth Elements, which are pictured in subsets. Depending on the type of periodic table, some columns may be numbered in Roman or Arabic numerals while others may use the number 1. The periodic table usually lists each element in a column and includes numerals indicating its weight and atomic number. For example, Hydrogen belongs to the Halogen family while the Alkali Metals belong to the group of non-metals. The first thing you should know about the periodic table is that most of the elements are organized into groups of one or two. Sodium ions have a positive charge, while chloride ions have a negative charge. The difference between protons and electrons is the ion’s net charge. One ion contains a positive charge, while the other has a negative charge. The periodic table shows that each element has both protons and electrons, forming the ion. A metal’s name usually ends in -ium, which is why so many of the elements on the periodic table end in this suffix. The ion is then formed through electrostatic attraction. They take the form of electrons or protons, and metals lose their electrons to make an ion. Ions are the particles that make up an element.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
